
Computational Optimization of a Stent for the Femoropopliteal Artery
link.springer.com
May 9, 2026, 9:57 a.m.
This study underscores the potential of multi-objective optimization in stent design, paving the way for PAD stents that more effectively accommodate femoropopliteal biomechanics and promote favorable mechanical conditions for healing.
Share on

Outcomes of drug-coated balloon combined with stenting vs. stenting alone in the treatment of femoropopliteal artery lesions requiring bailout stenting
link.springer.com
May 9, 2026, 9:55 a.m.
In patients requiring bailout stenting for FPA lesions, adjunctive DCB use was associated with lower 1-year CD-TLR; however, this finding should be considered hypothesis-generating due to the retrospective design and limited sample size. No sustained benefit was observed at 2 years. No signal for increased short-term mortality was identified, although the study was not powered for safety endpoints.
Share on

Cordis Selution SLR PTA Drug-Eluting Balloon Launched in Japan
evtoday.com
May 8, 2026, 9:27 p.m.
Cordis has launched the Selution SLR percutaneous transluminal angioplasty drug-eluting balloon in Japan for treating peripheral artery disease. The device delivers sirolimus in a controlled, sustained manner to reduce restenosis and restore vessel function. Clinical trial data from SELUTION SFA Japan demonstrated robust long-term outcomes, achieving 81.5% primary patency and 93.8% freedom from reintervention at three years in a complex patient population. Results were presented at the Japan Endovascular Treatment conference and published in JACC: Cardiovascular Interventions by principal investigators Yoshimitsu Soga and Osamu Iida.
Share on
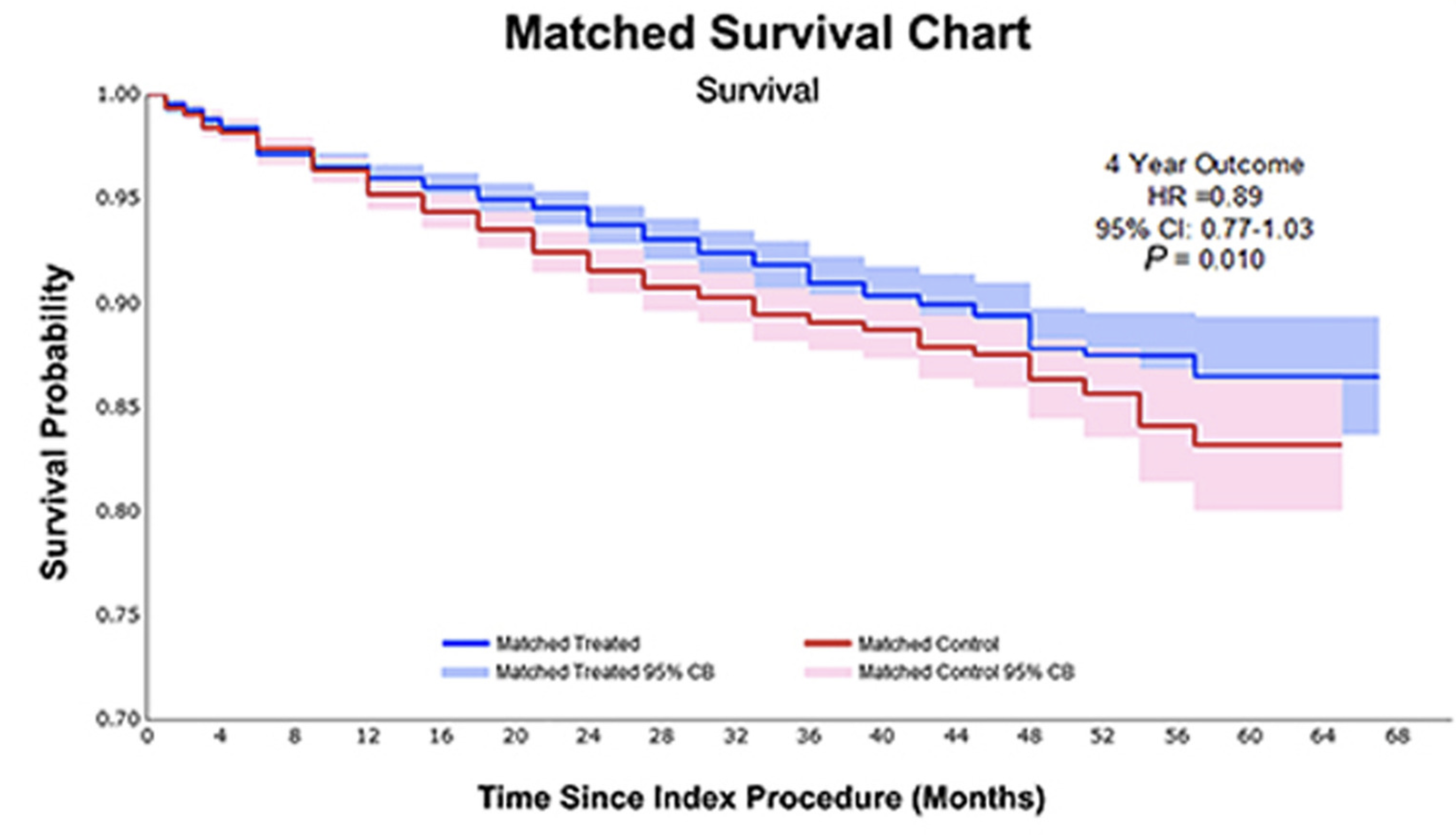
Safety of paclitaxel-coated devices in patients with peripheral artery ...
pmc.ncbi.nlm.nih.gov
May 8, 2026, 9:26 p.m.
This updated systematic review and meta-analysis examines the safety profile of paclitaxel-coated devices in peripheral artery disease treatment. Conducted by researchers from Nanjing Medical University, the study synthesizes evidence from randomized controlled trials to evaluate clinical outcomes and adverse events associated with these drug-coated interventional devices. The research contributes to ongoing clinical assessment of paclitaxel-eluting technology in vascular interventions, providing evidence-based insights for clinicians managing peripheral arterial disease. This comprehensive analysis addresses critical safety considerations essential for informed decision-making in peripheral vascular interventions and supports evidence-based clinical practice guidelines.
Share on

Trial Establishes Benefit of Drug-Eluting Stent in PAD
www.dicardiology.com
May 8, 2026, 9:23 p.m.
Drug-eluting stents are beneficial in treating symptomatic peripheral artery disease (PAD) in the femoropopliteal artery. Data from the ZILVER PTX trial demonstrate that paclitaxel-eluting stents had significantly better 12-month patency rates compared to traditional angioplasty with bare-metal stents for lesions in the femoropopliteal artery (above the knee).
Share on
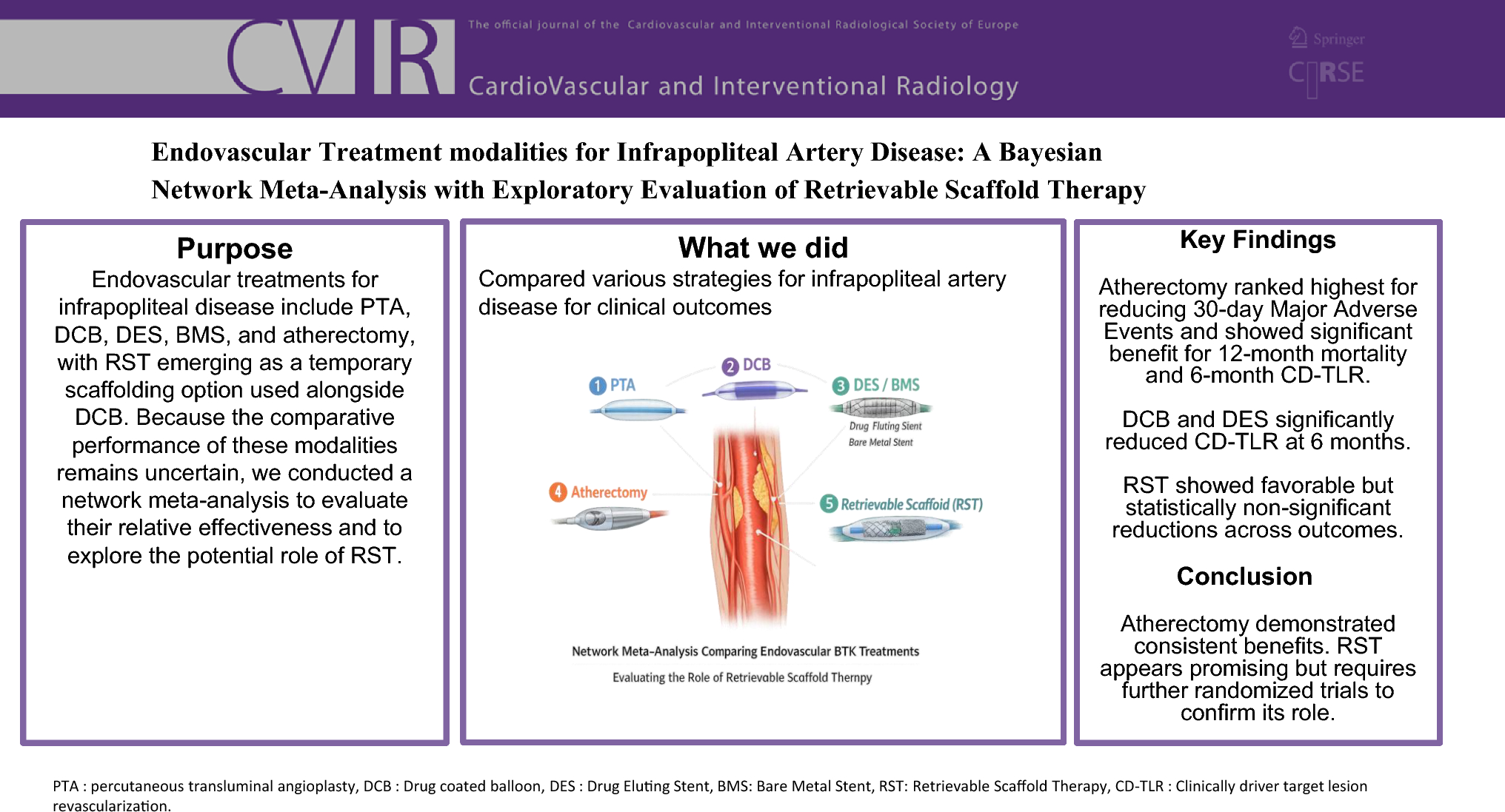
Endovascular Treatment Modalities for Infrapopliteal Artery Disease: A ...
link.springer.com
May 8, 2026, 8:32 p.m.
This comprehensive Bayesian network meta-analysis evaluates endovascular treatment options for infrapopliteal artery disease, including plain balloon angioplasty, drug-coated balloons, drug-eluting and bare-metal stents, and atherectomy, with exploratory assessment of retrievable scaffold therapy. The analysis encompassed 21 randomized and 4 single-arm trials involving 3,184 patients, measuring outcomes including 30-day major adverse events, 12-month mortality, 6-month target lesion revascularization, and major amputation rates. Results indicate atherectomy ranked superior for reducing 30-day major adverse events and demonstrated significantly lower 12-month mortality compared to plain balloon angioplasty. These findings provide valuable comparative effectiveness data to inform clinical decision-making in treating infrapopliteal artery disease.
Share on

CONCEPT MEDICAL ANNOUNCES PRELIMINARY 3-YEAR ...
www.tctmd.com
May 8, 2026, 8:32 p.m.
Concept Medical presented preliminary three-year data from the SIRONA randomized trial at the Charing Cross Symposium 2026, demonstrating the durability of its MagicTouch PTA sirolimus-coated balloon for treating femoropopliteal artery disease. The study compared sirolimus-coated versus paclitaxel-coated balloon angioplasty, showing superior freedom from clinically driven target lesion revascularization at three years with the sirolimus approach. The findings suggest that sirolimus-coated balloon angioplasty may provide more durable long-term outcomes than traditional paclitaxel alternatives, while maintaining comparable safety profiles with balanced all-cause mortality rates and low amputation rates across both treatment groups.
Share on

REVA Medical Announces Positive MOTIV BTK Trial Results Showing Superiority in Patients with Critical Limb-Threatening Ischemia
www.prnewswire.com
May 8, 2026, 5:45 p.m.
REVA Medical, LLC today announced primary endpoint results from the MOTIV BTK randomized pivotal clinical trial evaluating the MOTIV® sirolimus-eluting bioresorbable vascular scaffold in patients with critical limb-threatening ischemia (CLTI). The results were presented today at the Charing Cross International Symposium in London, UK. Motive Sirolimus- Eluting Bioresorbable Vascular Scaffold System. The MOTIV BTK trial met both its primary safety and efficacy endpoints, demonstrating that the MOTIV scaffold provides a statistically significant improvement in clinical outcomes compared to the current standard of care, balloon angioplasty.
Share on

Outcomes of drug-coated balloon combined with stenting vs. stenting alone in the treatment of femoropopliteal artery lesions requiring bailout stenting
link.springer.com
May 8, 2026, 2:33 p.m.
In patients requiring bailout stenting for FPA lesions, adjunctive DCB use was associated with lower 1-year CD-TLR; however, this finding should be considered hypothesis-generating due to the retrospective design and limited sample size. No sustained benefit was observed at 2 years. No signal for increased short-term mortality was identified, although the study was not powered for safety endpoints.
Share on
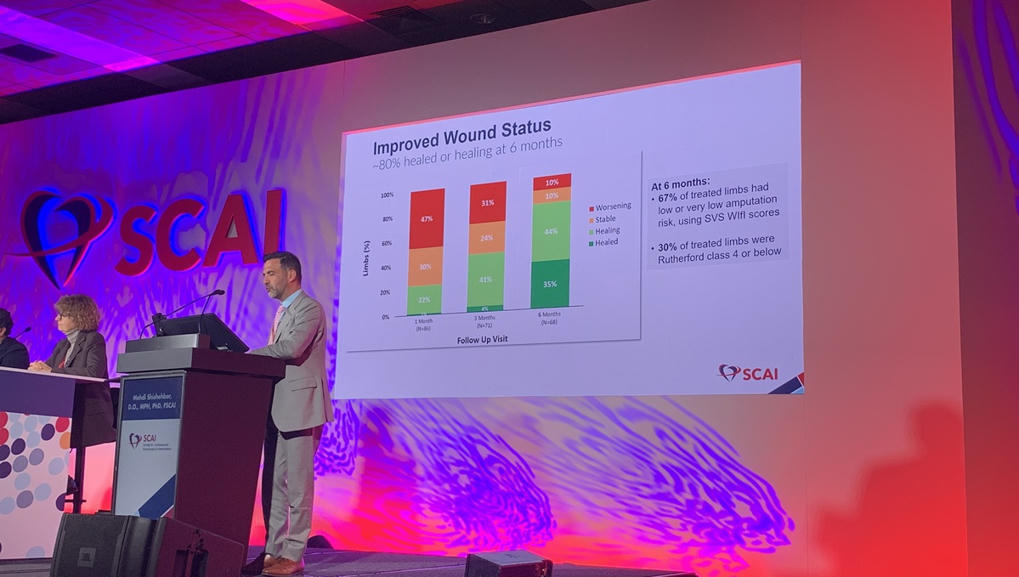
No-Option CLTI Patients Report Better QoL After Blood Flow Diversion Therapy: PROMISE III
www.tctmd.com
May 4, 2026, 2:05 p.m.
In patients with chronic limb-threatening ischemia (CLTI) who have run out of treatment options, a blood flow-diverting procedure in the foot improves several measures of quality of life (QoL) in addition to lowering the risk of major amputation, according to a secondary analysis of the PROMISE III trial.At 6 months, patients reported being less bothered about poor circulation in their legs compared with before the procedure, as well as more participation in social activities, better sleeping habits, and fewer daily burdens associated with having open wounds.
Share on

enVVeno Medical Receives FDA IDE Approval for Non-Surgical Replacement Venous Valve
medtechintelligence.com
May 4, 2026, 7:01 a.m.
enVVeno Medical has received the investigational device exemption (IDE) approval from the US Food and Drug Administration (FDA) for a pivotal study involving a non-surgical replacement venous valve. The approval allows enVVeno Medical to begin the Transcatheter Venous Valve Endoprosthesis (TAVVE) pivotal study of its enVVe system for treating severe deep chronic venous insufficiency (CVI).
Share on
FDA denies PMA submission for enVVeno Medical's VenoValve
www.massdevice.com
May 4, 2026, 7 a.m.
The FDA’s letter indicates that, upon completing its review, it determined that it is unable to approve the PMA for VenoValve in its current form. According to a news release, the FDA indicated that the favorable revised Venous Clinical Severity Score (rVCSS) data generated by the study to show clinical improvement, together with the improvements in pain scores and venous-specific quality of life indicators, were not sufficient on their own to determine the favorability of the risk-benefit profile for VenoValve.EnVVeno Medical said that, without a specific hemodynamic measurement that correlates with patient improvement, the FDA raised concerns about bias. The agency also considered the possibility that clinical improvement occurred as a result of the patients being enrolled in a study.Additionally, the company said the FDA focused on safety concerns attributed to the associated open surgical procedure. That required rehospitalizations, the company said. However, it would not expect to see similar safety events with a non-surgical replacement valve.
Share on

Interim results of covered stent-graft hint at “new avenue” in dialysis access treatment
vascularnews.com
April 26, 2026, 6:51 p.m.
“The reason for stent graft failure is neointernal hyperplasia that occurs mainly on the edge of the device and sometimes in the middle of the stent graft,” Harduin detailed. The Solaris DE covered stent is built with an impermeable electrospinning PTFE membrane to limit cellular migration, allied with the sirolimus coating to block neointimal hyperplasia and restenosis. “Sirolimus is a safe and effective cytostatic drug that has been used for a long time in interventional cardiology in coronary arteries to prevent neointimal hyperplasia, restenosis, and thrombosis,” said Harduin.Animal studies of Solaris DE have, to date, shown the persistence of the sirolimus on the outer edge of the device, he said, with one study comparing it to a conventional stent graft finding no instances of neointimal hyperplasia at 60 days following Solaris DE implantation, compared to 4.9mm3 with the conventional device.
Share on

Strutless bioresorbable BTK scaffold demonstrates technical success and primary safety in first cases
vascularnews.com
April 26, 2026, 6:42 p.m.
The RFS device is a bioresorbable stent built from microfibres, providing structural support to instantly open, and facilitate the reconstruction of the artery. Due to the porous design of the implant, patients’ own cells infiltrate into the mesh, triggering the formation of new vascular tissue. While the artery is being reconstructed from the inside-out, the synthetic implant gradually resorbs and ultimately disappears over time.At LINC, Brodmann expanded on some key characteristics of the RFS. “The device is a fully electrospun tubular conduit with fibrous micro-architecture and a strutless design,” the presenter noted, also highlighting the implant’s suitability for transcatheter delivery due to a balloon-expandable design.
Share on

BARISTA trial demonstrates positive one-year results in treatment of illac artery lesions
vascularnews.com
April 26, 2026, 6:41 p.m.
The BARISTA trial, led by Lieven Maene (Onze Lieve Vrouw, Aalst, Belgium), enrolled 200 patients with all types of iliac lesions. Significantly, 34% of the study population presented with TASC C and D lesions, representing more complex anatomical scenarios and underscoring the robustness of the clinical evaluation.At one-year follow-up, the trial demonstrated positive clinical outcomes: Freedom from target lesion revascularisation (fTLR): 96.4% Freedom from restenosis: 94.1%
Share on
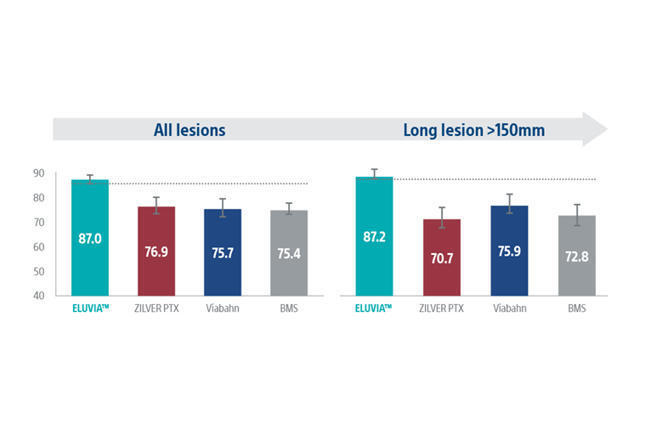
“Impressive” meta-analysis results at two years for Eluvia drug-eluting stent
vascularnews.com
April 26, 2026, 6:39 p.m.
Launched in Europe in 2016 and the USA two years later following successful results from the IMPERIAL randomised controlled trial (RCT),1 the Eluvia DES is designed to provide sustained drug release and efficient drug transfer with the aim of overcoming the challenge of restenosis in the superficial femoral artery (SFA).Steiner’s clinical experience with the Eluvia DES dates back several years. At this time, she and colleagues conducted the BEST-SFA RCT, in which Eluvia was used as a comparator. “What we observed in this analysis, and in our own clinical experience, was that the stent performed very well, effectively inhibiting the need for reintervention.” Steiner recalls. Two-year results of this study were published in 2024 in JACC: Cardiovascular Interventions.2
Share on

LUMIFOLLOW registry shows sustained three-year performance of Luminor in femoropopliteal disease
vascularnews.com
April 26, 2026, 6:31 p.m.
LUMIFOLLOW is a multicentre, real-world registry including 542 patients from 15 centres in France. The study reflects routine clinical practice and includes a broad spectrum of lesion complexity. Notably, 43.6% of lesions were classified as TASC C and D, and 24.2% of treated cases were restenotic lesions, underlining the challenging nature of the population evaluated.At three years, 87.3% of patients remained free from revascularisation, confirming durable treatment effect, states the company. No device-related safety concerns were reported during the follow-up period.
Share on

Spur RST: a novel approach to address limitations in BTK CLTI
vascularnews.com
April 8, 2026, 8:29 p.m.
Retrievable scaffold therapy (RST) with the Spur® peripheral retrievable scaffold system (Reflow Medical; Figure 1) represents a novel approach to BTK CLTI treatment. The Spur system consists of a retrievable nitinol scaffold with radial spikes integrated onto a balloon delivery platform. The radial spikes are designed to penetrate the lesion and vessel wall in a controlled manner, reduce recoil, change vessel compliance, and create channels that enhance drug uptake to the adventitia (Figure 2).15 Following Spur RST, commercially available DCBs are applied to provide long-term patency.16–18 This combined strategy is intended to deliver stent-like results while leaving nothing behind.
Share on

SirPAD: Primary Results confirm the safety and efficacy of Sirolimus Coated Balloon in PAD
www.prnewswire.com
April 6, 2026, 10:29 a.m.
The academic, all-comers SirPAD trial enrolled 1,252 patients with femoropopliteal or below-the-knee PAD who were randomized to receive either the sirolimus-coated balloon MagicTouch PTA or any uncoated balloon angioplasty. The primary outcome of major adverse limb events (MALE), encompassing major unplanned amputations affecting the target limb or target-lesion revascularization for critical ischemia, occurred in 8.8% of patients enrolled in the sirolimus-coated balloon group vs. 15% in the uncoated balloon group at one year, corresponding to a median unbiased estimate of the risk difference of -4.9%. This difference was both noninferior and superior in favour of MagicTouch PTA (Concept Medical Inc.) vs. uncoated balloon. Furthermore, the authors showed a statistically significant reduction in the composite key secondary endpoint of any unplanned target-limb amputation or any target limb revascularization.
Share on

SUCCESS PTA: 12-Month Data from SELUTION SLR™ DEB
www.radcliffevascular.com
Dec. 15, 2025, 8:37 a.m.
12 Month Outcomes of the SUCCESS PTA Study show consistent safety and effectiveness in claudicant and CLTI patients when using the SELUTION SLR™ drug eluting balloon.
Share on