
Wave propagation in a model artery
comptes-rendus.academie-sciences.fr
May 10, 2026, 10:37 a.m.
Dans cet article, nous étudions expérimentalement la propagation d’ondes dans un modèle d’artère : un ruban d’élastomère couplé à un canal rigide. Nous mesurons les ondes hors plan par imagerie Schlieren synthétique, et mettons en évidence un unique mode dispersif, semblable à l’onde de pouls générée par les battements du cœur. En imposant une différence de pression hydrostatique, nous révélons la forte influence de la précontrainte sur la dispersion de cette onde. À l’aide d’un modèle fondé sur la théorie acoustoélastique, prenant en compte la rhéologie du matériau et la grande déformation statique du ruban, nous démontrons que la pression imposée altère la propagation des ondes par une interaction entre l’étirement, perpendiculaire à la direction de propagation, et la rigidité induite par la courbure.
Share on
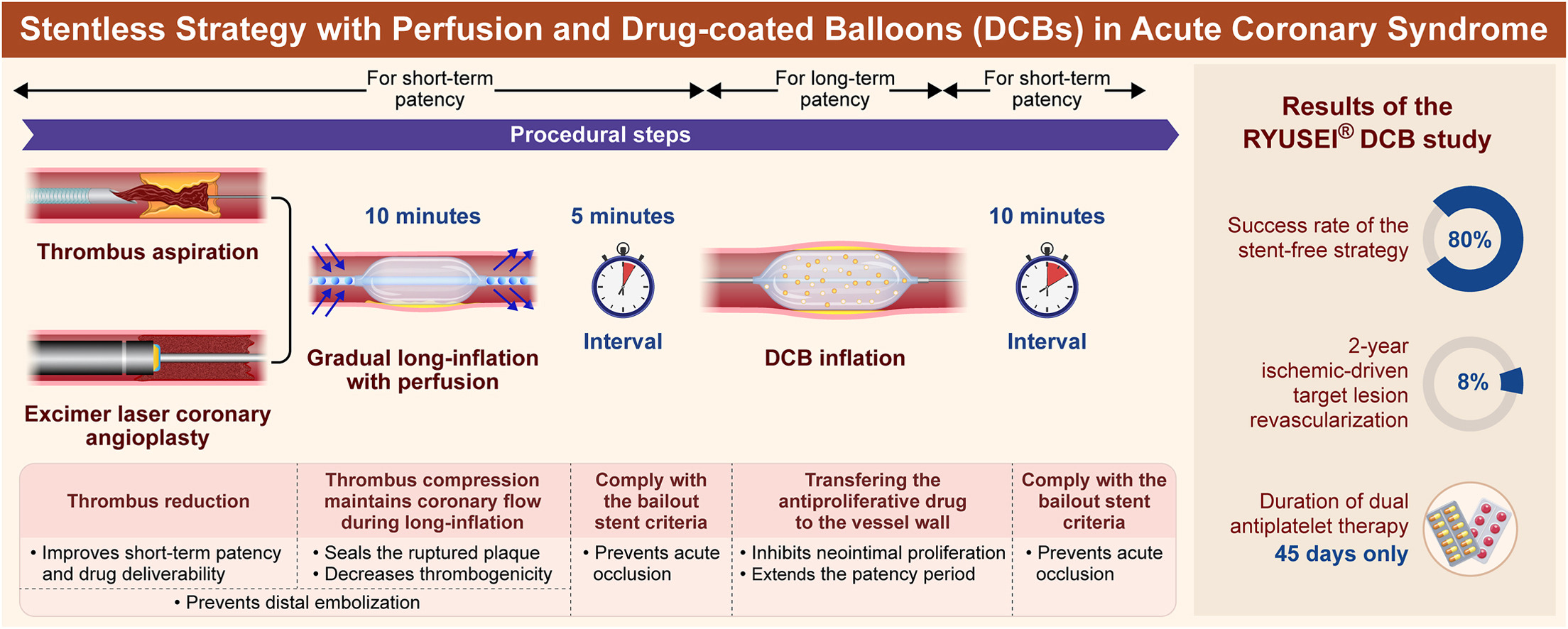
Novel Stentless Strategy With Perfusion and Drug-Coated Balloons for Treating Acute Coronary Syndrome
www.jscai.org
May 9, 2026, 10:10 a.m.
Stentless treatment of acute coronary syndrome uses gradual, prolonged predilation. A perfusion balloon combined with a drug-coated balloon was used. The stentless strategy achieved successful revascularization in most patients. No acute occlusion or in-hospital major adverse cardiac events were reported. A low incidence of target vessel failure at 24 months was reported.
Share on
Clinical Outcomes of Bail-Out Stenting After Drug-Coated Balloon Angioplasty: The International Multicenter BAILOUT Registry
www.ahajournals.org
May 9, 2026, 10:04 a.m.
In cases of suboptimal angiographic results after DCB angioplasty, bail-out DES implantation is safe, with no increased risk of TLF at 1 year compared with the expected performance goal for an upfront DES-only strategy.
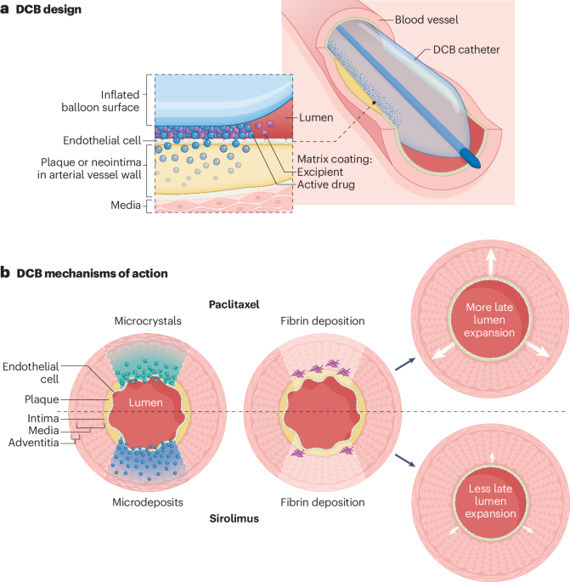
Drug-coated balloon angioplasty for coronary and peripheral artery disease: latest evidence and clinical indications
www.nature.com
May 9, 2026, 10:01 a.m.
Drug-coated balloon (DCB) angioplasty provides an alternative to drug-eluting stents and plain old balloon angioplasty for the treatment of coronary or peripheral artery disease. The absence of a metallic scaffold with DCB angioplasty compared with stenting might confer a biomechanical and physiological benefit and avoids the implantation of additional stent layers in patients with in-stent restenosis. A class effect cannot be assumed for DCBs; device manufacturers are challenged with finding the best combination of antiproliferative agent and excipient to achieve optimal clinical and angiographic outcomes. Several randomized clinical trials have been performed comparing DCB angioplasty with a variety of comparators in both coronary and peripheral artery disease, although more evidence is needed, particularly in de novo coronary artery disease. Positive results from a trial comparing a paclitaxel-coated balloon with an uncoated balloon in patients with coronary in-stent restenosis led to the approval of a DCB for clinical use in the USA in 2024.
Share on

Novel device therapies in heart failure: focus on patient selection
www.frontiersin.org
May 9, 2026, 9:33 a.m.
The increasing prevalence of heart failure (HF) has led to advancements in therapeutic strategies, including the development of new pharmacological treatments and the expansion of guideline recommendations across the spectrum of left ventricular ejection fractions. Despite these advancements, the full benefits of guideline-directed medical therapy (GDMT) are often limited by various barriers that result in incomplete implementation or suboptimal responses. For patients who cannot tolerate or only partially respond to GDMT, therapeutic options remain limited. This gap is particularly significant for those with contraindications to heart replacement therapies (HRT), such as left ventricular assist device (LVAD) or heart transplant. In light of these potential limitations, this review article proposes categorizing HF patients into four distinct phenoprofiles based on their tolerance to GDMT and candidacy for HRT. Considering these HF phenoprofiles may guide treatment decisions regarding the selection and use of novel device-based HF therapies.
Share on

Slow flow and no reflow after percutaneous coronary intervention
eurointervention.pcronline.com
May 9, 2026, 9:31 a.m.
Primary percutaneous coronary intervention is currently the standard of care for the treatment of acute ST-segment elevation myocardial infarction (STEMI). While it can restore flow in the infarcted artery in the majority of cases, some patients experience the so-called “no-reflow” phenomenon, which consists of abnormal myocardial reperfusion occurring after the occluded coronary artery has been opened. It is caused by microvascular obstruction (MVO), and its pathogenesis is multifactorial, including myocardial ischaemia, distal embolisation, and ischaemia-reperfusion injury, combined with individual susceptibility. Currently, there is no consensus on pharmacological or interventional strategies which can prevent or treat it.
Share on

Comparison of percutaneous Intervention with Drug Coated balloons and drug Eluting stents for de novo coronary artery disease: CIRCE, a systematic review and meta-analysis
www.sciencedirect.com
May 8, 2026, 10:23 p.m.
No significant difference was observed between DCB and DES regarding major adverse cardiovascular events (MACE), mortality, myocardial infarction, or repeat revascularization on a mean follow-up of 2 years (6 months to 5 years). DCB significantly reduced BARC 3-5 bleedings compared with DES (RR 0.65; 95% CI 0.43–0.98; p = 0.04). The findings were consistent across vessel sizes and follow-up durations. DCB therapy represents a safe and effective “leave-nothing-behind” strategy for selected patients undergoing PCI.
Share on

Drug coated balloons in percutaneous coronary intervention: how can computational modelling help inform evolving clinical practice?
www.frontiersin.org
May 8, 2026, 10:22 p.m.
Despite the growing interest in DCB within the clinical community, the mechanisms of drug exchange and the interactions between the balloon, the polymeric coating and the vessel wall are yet to be fully understood. It is, therefore, perhaps surprising that the number of computational (in silico) models developed to study interventions involving these devices is small, especially given the mechanistic understanding that has been gained from computational studies of DES procedures over the last two decades. In this paper, we discuss the current and emerging clinical approaches for DCB use in PCI and review the computational models that have been developed thus far, underlining the potential challenges and opportunities in integrating in silico models of DCB into clinical practice.
Share on
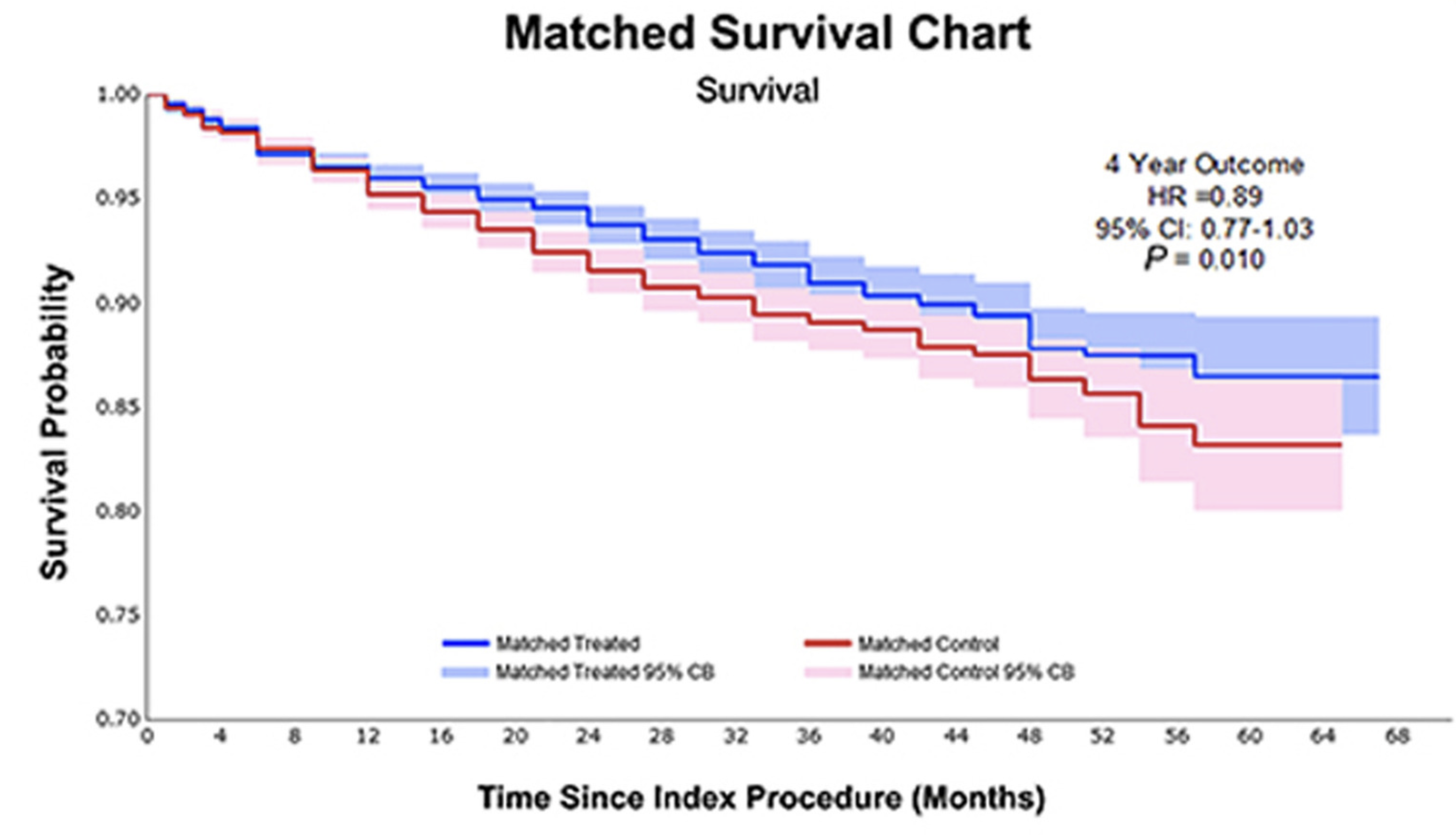
Safety of paclitaxel-coated devices in patients with peripheral artery disease
pmc.ncbi.nlm.nih.gov
May 8, 2026, 9:26 p.m.
This systematic review and meta-analysis examines the safety profile of paclitaxel-coated devices in treating peripheral artery disease by synthesizing data from randomized controlled trials. Paclitaxel-coated balloons and stents have emerged as promising interventional tools for managing peripheral vascular disease, yet concerns regarding their long-term safety persist. The study provides an updated evidence synthesis to clarify the safety implications of these drug-eluting devices in peripheral applications. By comprehensively analyzing randomized trial data, this research contributes critical insights into adverse event rates, mortality outcomes, and clinical efficacy, informing clinical decision-making for interventional specialists managing peripheral artery disease patients.
Share on

Trial Establishes Benefit of Drug-Eluting Stent in PAD
www.dicardiology.com
May 8, 2026, 9:23 p.m.
Drug-eluting stents are beneficial in treating symptomatic peripheral artery disease (PAD) in the femoropopliteal artery. Data from the ZILVER PTX trial demonstrate that paclitaxel-eluting stents had significantly better 12-month patency rates compared to traditional angioplasty with bare-metal stents for lesions in the femoropopliteal artery (above the knee).
Share on

Solaris DE Drug-Eluting Covered Stent Receives FDA Breakthrough ...
evtoday.com
May 8, 2026, 6:48 p.m.
Solaris Endovascular has received FDA Breakthrough Device designation for its Solaris DE drug-eluting covered stent, a significant advancement in vascular intervention technology. The device is specifically designed to treat stenosis in hemodialysis patients at the venous anastomosis of synthetic arteriovenous grafts. By combining a self-expanding nitinol scaffold with an impermeable polytetrafluoroethylene barrier and targeted sirolimus elution, the Solaris DE directly addresses edge restenosis, a leading cause of stent failure in dialysis access and other vascular applications. Preliminary data from the DEScover clinical trial demonstrated impressive results, achieving 100% target lesion primary patency at six months in the arteriovenous graft cohort, supporting the FDA's expedited designation pathway.
Share on

IN.PACT AV access post-approval study: Outcomes from extended ...
renalinterventions.net
May 8, 2026, 5:48 p.m.
The IN.PACT arteriovenous access post-approval study presented extended cohort outcomes at the 2026 Charing Cross Symposium, demonstrating clinical results consistent with prior experience. This FDA-mandated prospective multicentre study evaluated the IN.PACT drug-coated balloon for treating arteriovenous circuit stenosis in end-stage kidney disease patients. The extended cohort included 99 patients with both fistulas and grafts, broadening beyond the initial 186-patient primary cohort. The study met its primary endpoint of serious infection rate at 12 months, achieving the 30 percent performance goal. Results showed higher infection rates in arteriovenous graft patients but remained favorable compared to existing literature. While twelve-month survival data aligned with expected outcomes, investigators noted that greater lesion heterogeneity and small sample sizes warrant cautious interpretation of findings.
Share on
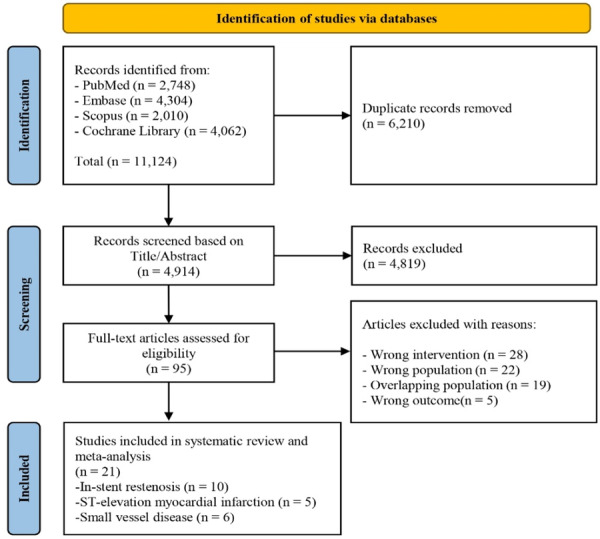
Outcomes of Drug‐Coated Balloon Versus Drug‐Eluting Stent for In ...
pmc.ncbi.nlm.nih.gov
May 8, 2026, 5:47 p.m.
This meta-analysis of randomized controlled trials compares the clinical outcomes of drug-coated balloons and drug-eluting stents for treating in-stent restenosis and de novo coronary lesions. The study, conducted by researchers at Tehran Heart Center, synthesizes evidence from multiple trials to evaluate the efficacy and safety profiles of these two interventional cardiology approaches. By analyzing randomized controlled trial data, the research provides valuable insights into treatment effectiveness, restenosis rates, and adverse event profiles. These findings are significant for clinical decision-making in coronary intervention strategies and help establish evidence-based guidance for selecting between these competing therapeutic modalities in different patient populations and lesion characteristics.
Share on
Drug-Coated Balloons versus Drug-Eluting Stents for Coronary ...
www.frontiersin.org
May 8, 2026, 5:47 p.m.
This systematic review and meta-analysis examines the comparative efficacy of drug-coated balloons (DCB) versus drug-eluting stents (DES) for treating coronary artery disease. Analyzing 23 randomized controlled trials involving 8,123 patients, researchers assessed major adverse cardiac events, target lesion revascularization, and various clinical outcomes across different lesion types and presentations. Results indicate that DCB showed higher rates of target lesion revascularization and composite endpoints compared to DES. The analysis includes lesion-specific stratification examining de novo lesions versus in-stent restenosis, vessel sizes, and clinical presentations including STEMI and NSTEMI, while also differentiating between paclitaxel and sirolimus-coated balloons. These findings provide evidence-based insights for clinical decision-making regarding revascularization strategy selection in coronary artery disease management.
Share on

What is a good introduction to drug‑eluting technology for peripheral ...
www.droracle.ai
May 8, 2026, 5:47 p.m.
# Summary
Drug-eluting technology represents a significant advancement in peripheral vascular interventions, enabling localized therapeutic drug delivery directly to target tissues. This innovative approach involves coating medical devices such as stents and balloons with pharmaceutical agents that gradually release medication to inhibit restenosis and promote vessel healing. By delivering drugs directly at intervention sites, this technology reduces systemic side effects while maximizing therapeutic efficacy. Drug-eluting devices have demonstrated improved clinical outcomes in peripheral artery disease treatment, offering enhanced durability and reduced reintervention rates compared to conventional bare-metal alternatives. This comprehensive introduction explores the fundamental principles, mechanisms, and clinical applications of drug-eluting technology in peripheral interventions, providing essential knowledge for medical professionals and interventionalists seeking to optimize patient outcomes.
Share on

FDA Clears Optical Coherence Tomography System for AI-Enabled High-Resolution Imaging of Coronary Plaque
www.diagnosticimaging.com
May 8, 2026, 2:35 p.m.
Facilitating enhanced real-time visualization for percutaneous coronary intervention (PCI) procedures, the Ultreon 3.0 optical coherence tomography platform reportedly combines high-resolution imaging with AI-powered insights on stent sizing and positioning.
Share on

Outcomes of Drug-Coated Balloon Versus Drug-Eluting Stent for In-Stent Restenosis and De-Novo Lesions
onlinelibrary.wiley.com
May 8, 2026, 2:30 p.m.
Drug-coated balloons (DCBs) have emerged as a promising device option in percutaneous coronary interventions and have been recently given FDA approval. In this study, DCB demonstrated comparable clinical outcomes with drug-eluting stent (DES) in patients with ISR and de novo lesions, including STEMI and SVD, except for lower LLL following DCB compared to DES in patients with SVD. Future trials are warranted to compare the clinical outcomes of DCB with DES in complex clinical settings like bifurcation or chronic total occlusion lesions.
Share on
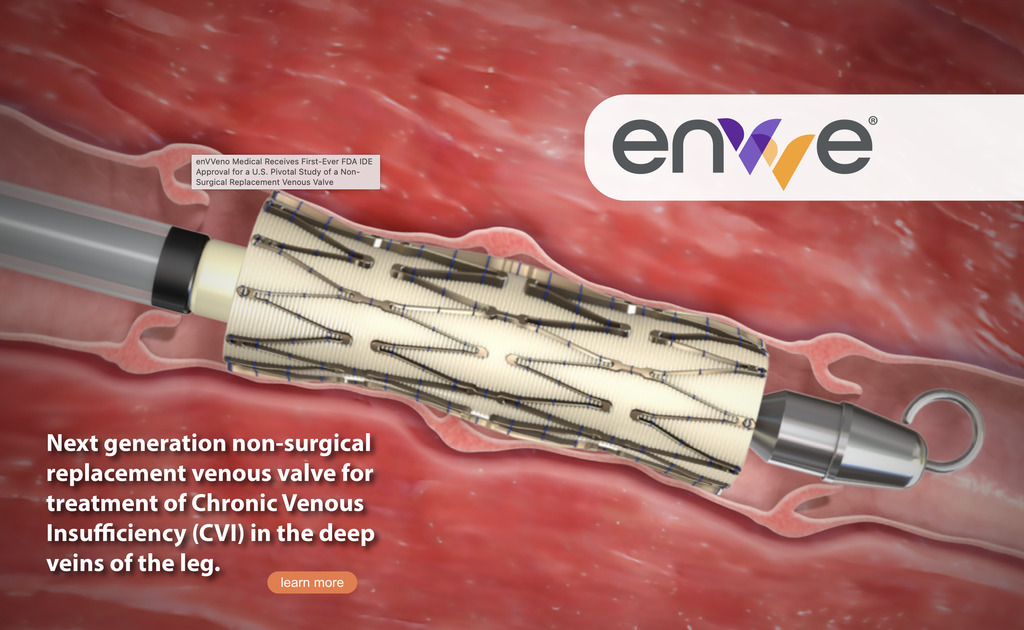
enVVeno Medical Receives FDA IDE Approval for Non-Surgical Replacement Venous Valve
medtechintelligence.com
May 4, 2026, 7:01 a.m.
enVVeno Medical has received the investigational device exemption (IDE) approval from the US Food and Drug Administration (FDA) for a pivotal study involving a non-surgical replacement venous valve. The approval allows enVVeno Medical to begin the Transcatheter Venous Valve Endoprosthesis (TAVVE) pivotal study of its enVVe system for treating severe deep chronic venous insufficiency (CVI).
Share on
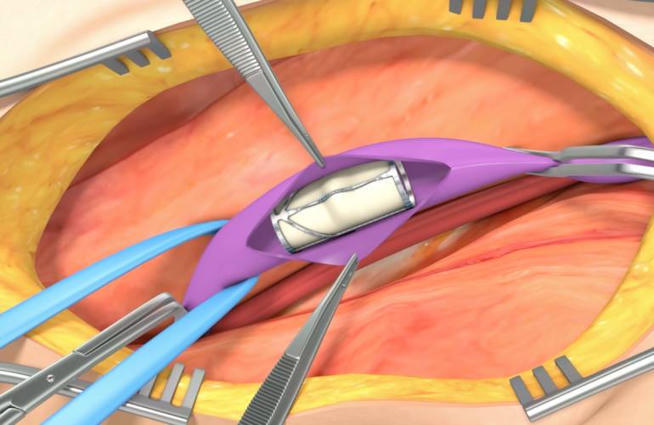
FDA denies PMA submission for enVVeno Medical's VenoValve
www.massdevice.com
May 4, 2026, 7 a.m.
The FDA’s letter indicates that, upon completing its review, it determined that it is unable to approve the PMA for VenoValve in its current form. According to a news release, the FDA indicated that the favorable revised Venous Clinical Severity Score (rVCSS) data generated by the study to show clinical improvement, together with the improvements in pain scores and venous-specific quality of life indicators, were not sufficient on their own to determine the favorability of the risk-benefit profile for VenoValve.EnVVeno Medical said that, without a specific hemodynamic measurement that correlates with patient improvement, the FDA raised concerns about bias. The agency also considered the possibility that clinical improvement occurred as a result of the patients being enrolled in a study.Additionally, the company said the FDA focused on safety concerns attributed to the associated open surgical procedure. That required rehospitalizations, the company said. However, it would not expect to see similar safety events with a non-surgical replacement valve.
Share on
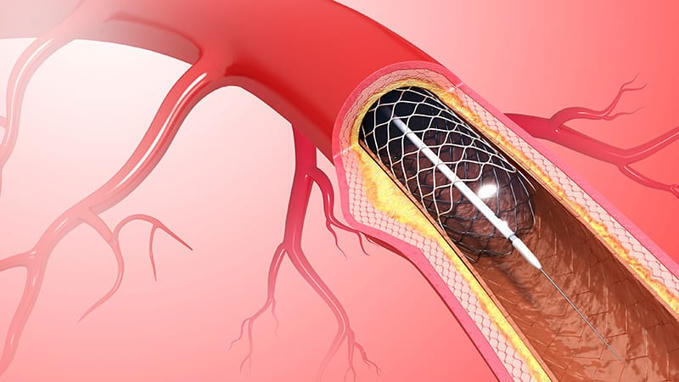
Sirolimus-Eluting Balloon Shows Promise in ACS
www.medscape.com
May 4, 2026, 6:53 a.m.
Patients with non-ST-elevation myocardial infarction (NSTEMI) or unstable angina undergoing a percutaneous coronary intervention (PCI) with a sirolimus-eluting balloon and rescue stenting fared as well as those who underwent revascularization with systematic use of drug-eluting stents at 1 year, according to a new substudy of the SELUTION DeNovo trial.
Share on