
Efficacy and safety of a new mechanical balloon-based flow diverter in the treatment of intracranial aneurysms
jnis.bmj.com
May 8, 2026, 10:40 p.m.
The mechanical balloon-based FD showed a remarkable occlusion rate alongside minimal ischemic and hemorrhagic adverse events compared with existing FDs. This innovative mechanical balloon-based design may be an important direction for future FD design.
Share on
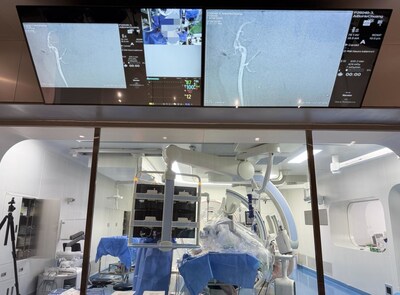
PANVIS STAR Vascular Interventional Robotic System Redefines ...
finance.yahoo.com
May 8, 2026, 10:19 p.m.
The PANVIS STAR Vascular Interventional Robotic System represents a significant advancement in treating ischemic stroke through mechanical thrombectomy. With stroke affecting millions globally and ischemic stroke comprising over 65% of cases, the demand for effective interventional solutions is critical. This robotic system addresses key challenges including limited patient access, operator fatigue, and steep learning curves by enabling remote-controlled procedures with enhanced precision. Demonstrated through successful animal model testing, PANVIS STAR successfully executed complete thrombectomy workflows including femoral access, vessel navigation, and clot retrieval. The system's innovative four-device co-manipulation capability, sub-millimeter precision, and haptic feedback enable operators to achieve superior outcomes while reducing the technical burden on healthcare professionals, potentially expanding access to this life-saving treatment globally.
Share on

Consensus statement seeks to standardise evaluation of robotic ...
neuronewsinternational.com
May 8, 2026, 10:19 p.m.
An international consensus statement has been published in the Journal of the American Heart Association establishing the first standardized framework for designing, testing, and evaluating robotic systems used in stroke thrombectomy. Led by King's College London, the statement addresses a critical gap in the field, as previous studies employed inconsistent methodologies, tested different tasks, and measured disparate outcomes, preventing meaningful comparisons. The international expert panel, comprising specialists in interventional neuroradiology, robotics, data science, health economics, policy, and patient advocacy, developed comprehensive guidelines to ensure patient safety while facilitating remote stroke treatment procedures. This standardization is expected to accelerate the advancement of robotic thrombectomy technology and expand access to emergency stroke care.
Share on
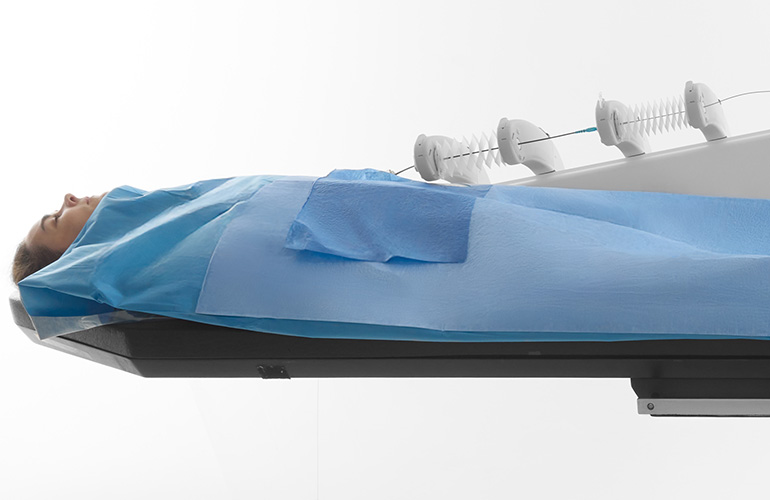
Sentante's breakthrough telesurgery system could redefine stroke care
www.medicaldesignandoutsourcing.com
May 8, 2026, 10:19 p.m.
Sentante has developed an innovative telesurgery system that could transform acute ischemic stroke treatment globally. The Sentante Stroke System, which recently received FDA Breakthrough Device designation, enables remote thrombectomies through a haptic robotic bedside unit controlled by endovascular specialists from distant locations. A landmark transatlantic demonstration in November 2025 showcased a Florida-based neurosurgeon successfully performing a procedure on a cadaver in Scotland. This technology addresses critical gaps in stroke care access, particularly for remote hospitals lacking local thrombectomy capabilities or timely transfer options. By combining digital innovation with robotic precision, Sentante's platform aims to enhance patient outcomes, improve efficiency, and ensure equitable access to life-saving interventions while addressing workforce sustainability challenges in endovascular medicine.
Share on
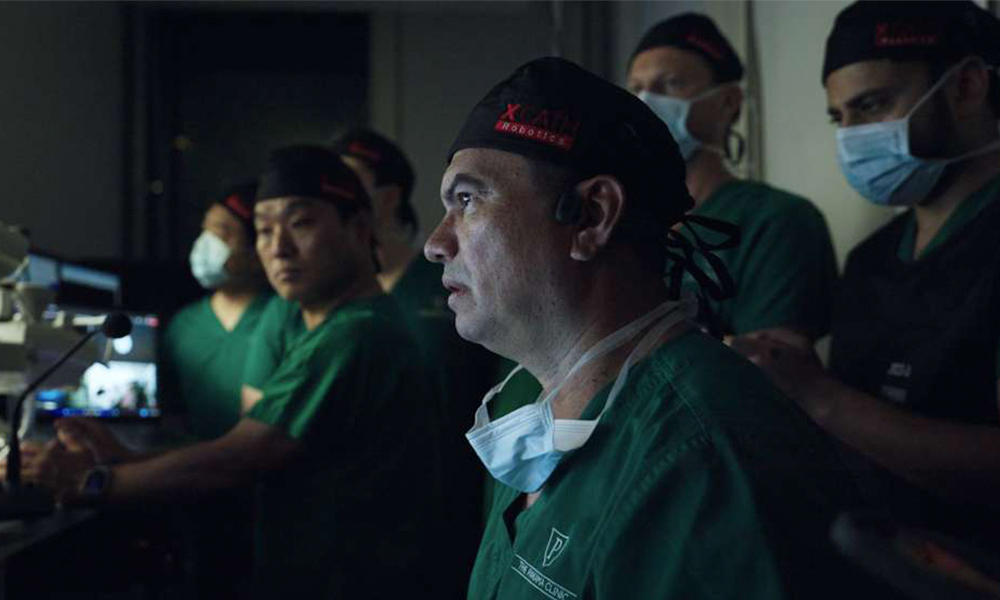
UAE-funded XCath completes world's first-in-human telerobotic stroke ...
www.crescententerprises.com
May 8, 2026, 10:19 p.m.
UAE-funded XCath, a neuroendovascular surgical robotics company partially owned by Crescent Enterprises, has successfully completed the world's first-in-human telerobotic stroke surgery. The landmark procedure involved a remote robotic mechanical thrombectomy performed using the XCath Iris Endovascular Robotic System, conducted by Dr. Vitor Mendes Pereira. This groundbreaking achievement represents a significant advancement in minimally invasive surgical technology and remote medical intervention capabilities, demonstrating the potential for telerobotic systems to enhance stroke treatment outcomes while expanding access to specialized surgical expertise across geographical distances.
Share on

Liberty - Microbot Medical
www.microbotmedical.com
May 8, 2026, 10:18 p.m.
Microbot Medical's LIBERTY® Endovascular Robotic System represents a transformative advancement in peripheral vascular intervention. This innovative single-use, remotely operated platform enables physicians to perform precise endovascular procedures from a seated position, significantly enhancing control and ergonomics. LIBERTY® eliminates the need for substantial capital investment while delivering a remarkable 92% reduction in radiation exposure compared to traditional manual techniques. The system integrates seamlessly into existing workflows with setup completed in under five minutes and maintains universal compatibility with standard guidewires and catheters. By combining compact design with intuitive remote operation, LIBERTY® modernizes vascular access and navigation, offering clinicians improved efficiency and peace of mind without the complexity associated with conventional capital equipment solutions.
Share on

Blood return system dramatically reduces blood loss during pulmonary thrombectomy
www.news-medical.net
April 27, 2026, 7:39 a.m.
The first comparative analysis from the ENGULF (A Safety and Feasibility Single-Arm Study of a Novel Catheter Thrombectomy Device For the Treatment of Pulmonary Embolism) trial shows that using blood return during continuous aspiration thrombectomy dramatically reduces blood loss by 97%, while maintaining hemodynamic efficacy and improving procedural efficiency. Researchers presented the late-breaking data today at the Society for Cardiovascular Angiography & Interventions (SCAI) 2026 Scientific Sessions & Canadian Association of Interventional Cardiology/Association Canadienne de cardiologie d'intervention (CAIC-ACCI) Summit in Montreal.
Share on

Albumin Plus Endovascular Therapy Boosts Stroke Outcomes
bioengineer.org
April 27, 2026, 7:38 a.m.
In a groundbreaking advancement for stroke therapy, a recent randomized clinical trial published in Nature Communications brings to light the synergistic effects of albumin administration combined with endovascular therapy for treating acute ischemic stroke patients. Acute ischemic stroke, a leading cause of disability and mortality worldwide, results from an abrupt obstruction of cerebral blood flow, often eliciting irreversible brain damage if not rapidly managed. While mechanical thrombectomy through endovascular techniques has revolutionized treatment by physically removing the occlusion, therapeutic adjuncts to enhance outcomes remain a critical unmet need. This study, led by Liu, Dong, Zhang, and colleagues, pioneers the exploration of albumin—a naturally abundant plasma protein—as a potential amplifier of neuroprotection and vascular restoration when paired with endovascular intervention.
Share on

Penumbra unveils latest iteration of thrombectomy system for venous thromboembolism
vascularnews.com
April 26, 2026, 6:55 p.m.
“Engineered for the rapid removal of pulmonary and venous thrombus, the latest Flash 3.0 algorithm is designed for enhanced clot detection capabilities with increased sensitivity to thrombus and blood,” Penumbra reported in a press release. The latest iteration includes enlarged tubing coupled with an automated backflush feature tailored for large thrombus burdens and to reduce friction from aspirated thrombus.The STORM-PE randomised controlled trial, recently published in Circulation, found that the use of Penumbra’s CAVT technology with anticoagulation achieved superior reduction in right heart strain compared to anticoagulation therapy alone in patients with acute intermediate-high risk pulmonary embolism (PE). This marks the first level-1 evidence demonstrating superior results for mechanical thrombectomy over traditional anticoagulation for the treatment of pulmonary embolism (PE).
Share on
E2 lands $80M series C for next-gen thrombectomy treatment
www.fiercebiotech.com
April 8, 2026, 8:54 p.m.
California-based medtech E2 (Endovascular Engineering) has raised $80 million in a series C funding round as it looks to commercialize its pulmonary embolism treatment. The Hēlo Thrombectomy Platform received FDA clearance late last year for use in peripheral veins to treat pulmonary embolism without the need for surgical intervention. The innovation behind the tech is its dual-action mechanism, which combines aspiration with mechanical clot disruption and is delivered through a small-profile catheter.
Share on

Wallaby Acquires German Neurovascular Leader Phenox to Accelerate Global Expansion
phenox.net
April 7, 2026, 9:29 a.m.
Wallaby, with its main offices in Laguna Hills, California and Shanghai, China achieves a significant portion of sales of its world-class and premier-quality products in the U.S. and other major developed markets. phenox, headquartered in Bochum, Germany, provides a broad portfolio of neurovascular devices to treat ischemic and hemorrhagic strokes that are sold in over 45 countries worldwide. The two companies have been strategic partners since 2019. phenox has been the exclusive distributor for Wallaby’s Avenir® Coil System in the U.S. and European markets as well as for Wallaby’s EsperanceTM Aspiration Catheters in the U.S. market.
Share on
David Ferrera: Turning Stroke Ideas Into Devices
www.intelligenthq.com
April 6, 2026, 10:33 a.m.
Big ideas are common in medicine. Turning them into real products is not. David Ferrera has spent nearly 30 years doing exactly that. He is an engineer, entrepreneur, and inventor based in Lake Forest, California. Over his career, he has helped build and sell multiple medical device companies. He holds more than 80 U.S. and international patents. He is also the author of Innovation in Translation: How Big Ideas Really Happen. But his focus is simple. “Innovation is important,” Ferrera says. “Translation is everything.”
Share on

Promising GPVI Inhibitor Advances Toward Clinical Development for Stroke
bioengineer.org
April 6, 2026, 10:32 a.m.
The molecule EMA601 precisely targets glycoprotein VI (GPVI), a platelet-specific receptor crucially implicated in thrombus formation and thrombo-inflammatory disease pathways. Unlike general antithrombotic agents that often interfere with primary hemostasis, EMA601 selectively inhibits pathological platelet activation without compromising the essential clotting functions necessary for safe hemostasis. This specificity stems from GPVI’s unique expression profile and its pivotal role in triggering platelet responses upon vascular injury, by binding exposed collagen and initiating intracellular signaling cascades.
Share on
MicroPort NeuroScientific granted US FDA breakthrough designation for Apollo Dream stent system
neuronewsinternational.com
April 6, 2026, 10:22 a.m.
As detailed in a company press release, intracranial atherosclerotic disease (ICAD) is a leading cause of ischaemic stroke worldwide and remains “one of the most challenging areas in cerebrovascular therapy”. Patients with medically refractory ICAD face limited treatment options, and effective interventional approaches to restore cerebral blood flow represent a “significant unmet clinical need”. To this end, Apollo Dream—MicroPort NeuroScientific’s investigational, balloon-expandable, rapid-exchange drug-eluting stent system—has been designed to provide vascular scaffolding and restore cerebral blood flow. The company says it is not aware of any other devices with this specific combination of features that have received a US FDA Breakthrough Device designation covering this indication.
Share on

NOVA intracranial DES re-opens CTO in left internal carotid artery C6 segment
mp.weixin.qq.com
April 3, 2026, 11:50 a.m.
The diameter of NOVA NEO stent covers 2.25mm to 4.00mm, and the length is 7mm to 20mm, which adapts to the needs of different blood vessel diameters and lesion lengths. The expansion of the balloon is completed synchronously with the release of the stent, which simplifies the surgical process and reduces the difficulty of operation. When passing through severe tortuous blood vessels, its flexibility, The passability performance is good, and the support for blood vessels after stent expansion is good, which can prevent the risk of stenosis of the stent after plaque retraction.
Share on
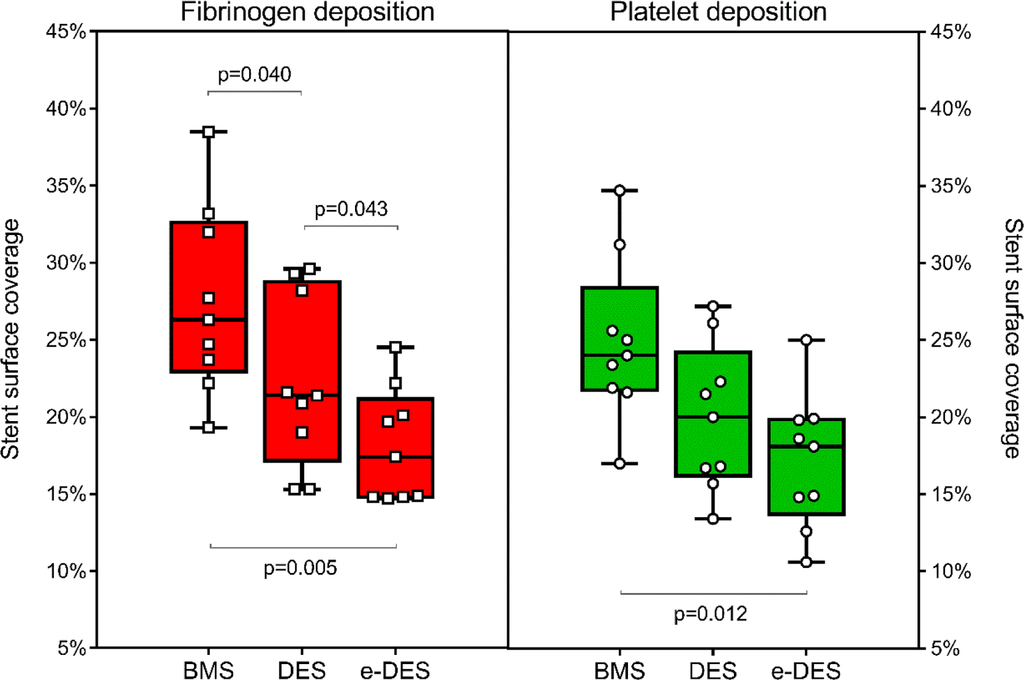
Effects of electret coating technology on coronary stent thrombogenicity
www.tandfonline.com
March 28, 2026, 2:17 p.m.
Stent thrombosis (ST) is a catastrophic event and efforts to reduce its incidence by altering blood-stent interactions are longstanding. A new electret coating technology that produces long-lasting negative charge on stent surface could make them intrinsically resistant to thrombosis. We assessed the thrombogenicity of stents using an annular perfusion model with confocal microscopy, and determined the efficacy of electret coating technology to confer thrombo-resistant properties to standard stents. Using an annular perfusion chamber, Bare Metal Stent (BMS), standard uncoated DES (DES), and Electret-coated DES (e-DES) were exposed to human blood under arterial flow conditions. Deposits of fibrinogen and platelets on the stent surface were analyzed using immunofluorescence staining and confocal microscopy. Surface coverage by fibrinogen and platelets and the deposit/aggregate size were quantified using computerized morphometric analysis.
Share on

Early Flow-Diverter Implantation in Ruptured Intracranial Aneurysms: Safety and Outcomes in a Resource-Limited Setting
neurointervention.org
March 16, 2026, 9:56 a.m.
Early FD implantation in carefully selected ruptured aneurysms, including small saccular and morphologically complex lesions can achieve high functional recovery and complete angiographic occlusion, even in a resource-limited environment. Ideal case selection and standardized DAPT and hemodynamic protocols are critical. These findings support broader use in challenging aneurysms, but larger prospective studies are warranted to validate outcomes and refine management strategies.
Share on
XCath secures US$30 million in Series C funding
neuronewsinternational.com
March 16, 2026, 9:55 a.m.
XCath has secured US$30 million in Series C funding, bringing the total raised since the company’s inception to US$92 million, with these latest funds set to support XCath’s ongoing efforts to bring “the world’s first commercially practical endovascular robot” to the market—and to perform a clinical telerobotic mechanical thrombectomy procedure.
Share on

Early Flow-Diverter Implantation in Ruptured Intracranial Aneurysms: Safety and Outcomes in a Resource-Limited Setting
neurointervention.org
March 11, 2026, 3:08 p.m.
Early FD implantation in carefully selected ruptured aneurysms, including small saccular and morphologically complex lesions can achieve high functional recovery and complete angiographic occlusion, even in a resource-limited environment. Ideal case selection and standardized DAPT and hemodynamic protocols are critical. These findings support broader use in challenging aneurysms, but larger prospective studies are warranted to validate outcomes and refine management strategies.
Share on

Optimization of braided stent design for cerebral aneurysms: the role of wire cross-sectional geometry
www.frontiersin.org
March 8, 2026, 5:04 p.m.
The overall blood flow distribution of stents with different cross-sections is similar, but there are differences in the local average flow velocity of the tumor cavity: the circular one is the highest, and the quadrilateral one is the lowest. (3) The wall pressure gradient change of the polygonal stent is gentler than that of the circular one. Among them, the wall pressure of the hexagonal and decagonal stents is the maximum and the average pressure in the cavity is the lowest. (4) The area of the low WSS region on the aneurysm wall is the largest in quadrilaterals and the smallest in circles. On the maternal artery segment, the hexagon is the largest and the quadrilateral is the smallest. Comprehensive comparison shows that quadrilateral and decagonal cross-section stents exhibit better comprehensive performance.
Share on